
Exhibition time: 17-19 March, 2026 Shanghai, China
 中文
中文

Exhibition time: 17-19 March, 2026 Shanghai, China
 中文
中文
Chelates are a hot new additive for soils that claim to make plants grow better and solve the problem of chlorosis. Chelated fertilizer use is also on the rise and even products that aren’t true chelates are reported to have chelating properties.
What is chelation (key-LAY-shun) and should you be adding chelates to soil? Are chelating properties good for plants?

What are Chelates?
In order to understand chelates you need to understand some things about the metal micronutrients such as iron, copper, zinc, magnesium and manganese. You may recognize some of these as being hard metallic substances, which they are. In order for plants to use them, the metal needs to be converted into a water soluble form called ions.
These ions float around in the water that surrounds soil particles. If they react with oxygen they are converted into a form that plants can’t use. They can also interact with other ions which may cause them to precipitate as solids, making them unavailable to plants.
This is the point in the story where chelates become important. Chelates are organic molecules (in the chemical sense) that pick up and hold the metal ions so that they don’t react with oxygen or other ions. Even though chelates hold on to the metal ions, plants can still absorb them through the roots and use them as nutrients.
Chelates make it easier for plants to find certain nutrients. In fact, plants use chelates internally to move metal ions from roots to leaves. Chelates are clearly important for plant growth.
Any chemical that has the properties described above has so-called chelating properties. Even glyphosate, the active ingredient in Roundup, acts like a chelate in soil.

Chelates Benefit Plants
The following are some of the ways chelates benefit plants (ref 1).
Make Ions Soluble
Some nutrient ions are less soluble in water than others and unless they are dissolved in water plants can’t use them. Chelation makes these ions more soluble, making them more accessible to plants.
Prevent Precipitation
At higher pH some metal ions, like iron, react with another ion called hydroxl (OH-). When these two ions join together, they are no longer soluble in water and precipitate (ie form a solid). This helps explains why some plants become chlorotic and show iron deficiency symptoms in alkaline soil even though the soil has lots of iron in it.
Chelation will reduce this reaction making more iron available to plants.
Reduce Toxicity of Metal Ions
Some metal ions become toxic to plants at higher concentrations. Chelation removes the ions from the water, reducing their concentration. Since plants are no longer exposed directly to the ion, the toxic effects are also reduced.
The chelating molecules responsible for this are usually large organic molecules that are commonly called humic acid.
Reduce Leaching
Chelates hold ions and keep them out of the soil water. Water running past the chelating molecules will no longer wash the ions away, reducing leaching. Chelates don’t completely eliminate leaching, but they certainly slow down the process.
Suppression of Plant Pathogens
By holding the ions, chelates make it more difficult for some pathogens to get access to them, thereby reducing their growth.

Sources for Chelates
Companies are now selling chelates in a powder form which you can add to soil. These products have ingredients such as, EDTA, DTPA and EDDHA.
You can also buy chelated fertilizer where the micronutrients are attached to chelate molecules during the manufacturing process. When the fertilizer is added to soil the ions are immediately protected from oxygen and other ions.
The natural source of chelates is organic matter. All organic matter has some degree of chelating properties and this is a very important role of organic matter in soil.
Plants Produce Chelates
Some plants like grasses will produce their own chelates in low-iron conditions. These non-protein amino acids are exuded by the roots into the soil solution surrounding the roots (the rhizosphere). These chelates pick up iron ions and hold onto them until they contact the root at which time they release the ion to the roots. The chelate molecule can then look around for another ion to repeat the process.
These chelates are acting like little taxis, driving iron molecules around until they bump into a root.
Select the Right Chelate
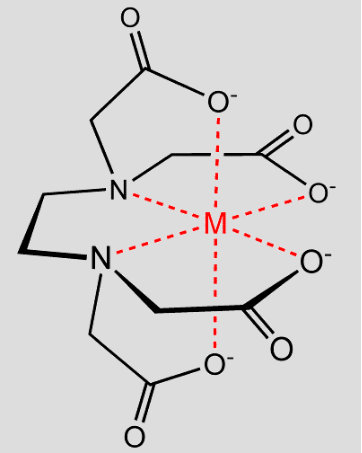
EDTA chelate showing the captured metal ion – “M”
Selecting the right commercial chelate fertilizer is important. Each chelate compound delivers a different amount of ion and is effective at different pH levels.
Iron – EDTA: releases the most iron to the soil, but it is only good in acidic soil. It is the common form found in most liquid fertilizers. EDTA also has a high affinity for calcium and should not be used on soil with a high calcium level since it becomes ineffective at protecting iron. EDTA works better with other metal ions at high pH.
Iron – DTPA: Effective up to a pH of 7.5 and not as sensitive to calcium as iron – EDTA.
Iron – EDDHA: Can be used up to pH 11, but it is the most expensive chelate in this group.
In addition to pH selecting the right chelate must also take into account the ion being delivered and the plant being fertilized. This is all much more complex than implied by product advertising, especially products geared towards home gardeners.
Have you ever seen a “suitable pH range” on a chelating product? Probably not. Once the consumer is convinced chelates are good for soil, they’ll by any chelating product regardless of their soil pH.
Chelates and the Environment
EDTA is one of the most popular chelates used in agriculture and unfortunately it is not quickly decomposed by microbes. It is also toxic to them. As a result there is an increasing amount of EDTA being found in our water systems.
EDTA on its own does not present much of a problem, but it also binds to heavy metals which makes them more available to plants. Accumulation of heavy metals in food crops can be a real problem (ref 3).
EDTA is also used in manufacturing which is a significant source of pollution.
Claims Made for Chelates
Plants Grow Better
This is true if plants are not growing well due to low nutrient levels but the effect of chelates in acidic soil is limited.
In agricultural situations, where the soil pH is alkaline, the addition of chelated fertilizer produces higher yields than adding regular fertilizer.
Stops Chlorosis
Iron deficiency produces interveinal chlorosis (also called iron chlorosis) not chlorosis. So adding chelated iron will not solve a chlorosis problem, as claimed for many products. Chlorosis can be the result of a number of problems, the most common being a lack of nitrogen, in which case chelates won’t help.
More details on this topic: Chlorosis in Plants – Is it Iron Deficiency
Good Foliar Feed
Some chelated products can be absorbed by the leaves in which case they will improve a nutrient deficiency. However, this will be a short term solution to the problem because the real problem in the soil has not changed.
Foliar feeding is not usually a good solution for home gardeners.
Should Home Gardeners Use Chelates?
There are three basic types of chelates; chelates without nutrients, chelated fertilizer and organic matter.
Chelates without nutrients are not a good idea for home gardeners since they don’t add the additional benefits of organic matter and cost more than organic matter.
Chelated fertilizer can be useful to solve specific problems. It might help growing acid-loving plants in alkaline soil. It could solve a nutrient deficiency problem. The biggest issue with this solution is that you need to know which deficiency you have, and then select the right kind of product. Home gardeners rarely have all of the details they need to make the right selection. Why not add organic matter instead?
Organic matter is always good for the garden. It will not only have chelating properties but it will also improve soil structure and increase water holding capability. This is the best way for gardeners to add chelates.
Glyphosate (Roundup) Chelates Ions
The glyphosate molecule functions like a chelate in soil, binding metal ions. This has led the anti-GMO/Roundup group to reach the conclusion that since it binds nutrients in soil, the plant must have fewer nutrients. Which leads them to the false conclusion that Roundup ready crops are less nutritious.
They ignore the fact that chelates actually make it easier for plants to find nutrients in the soil.
They also forget that Roundup ready crops also have lower weed competition allowing them to grow better. A recent review of this topic found that crops treated with Roundup are NOT less nutritious (ref 2).
References:
Chelated Fertilizers; http://edis.ifas.ufl.edu/hs1208
Glyphosate Effects on plant Mineral Nutrition; https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3479986/
EDTA – the Chelating Agent Under Environmental Scrutiny; http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-40422003000600020
